CEE-367: Aquatic Chemistry
Lecture
notes: Precipitation of Iron Hydroxides
OBJECTIVES:
-
Introduce
principles of coagulation for water treatment
-
Construct
and interpret logC-pH diagrams for solid-aqueous phase equilibrium
-
Solve
example problems with precipitation of solid phase
COAGULATION IN WATER TREATMENT:
Coagulation-a process for
combining small particles into larger aggregates.
Many water-borne pollutants of
concern to human health are either particles (pathogenic organisms) or
tend to associate with particulate materials due to low
solubility
(many organic compounds, toxic metals). A common method of removing
these
particles is to allow them to settle out, after "scavenging" them on
active
surfaces (see Chapter # 8). However, very small particles have settling
out times on the order of days or even weeks. Coagulation can reduce
the
time required to separate these particles by increasing the size and
mass
of the average particle, reducing their settling time. For this reason,
coagulation is an essential feature of most water treatment systems.
In order to overcome the stability of particles in treated water, a
coagulant
is added. This coagulant is a material that causes particles to
aggregate
into larger heavier masses (known as flocs) which can be more easily
removed
through settling or filtration. A common
treatment
process is to add metal salts in concentrations sufficient to cause
precipitation
of the metal hydroxide. These metal hydroxides are known as "sweep
flocs".
They collect colloidal particles both during formation of the
precipitate
and through subsequent collisions. In addition, dissolved organic
material
will adsorb to these precipitates, helping to remove color and odor
from
the water.
The most common metals used in coagulation are aluminium and iron(III),
as they both form insoluble metal hydroxides at low concentrations.
These
metals are usually added as sulfates or chloride species: for example,
FeCl3, Fe2(SO4)3, AlCl3,
and alum (Al2(SO4)3*14 H2O).
The exact dose required is a function of the chemistry of the treatment
water, particularly the pH, alkalinity, hardness, ionic strength, and
temperature.
The Iron Hydroxide System
-
When
iron(III)
is added to water, the Fe+3 ion reacts with OH-
to
form hydrolysis species and Fe(OH)3 (s).
-
Species:
(H2O), H+, OH-, Fe+3, FeOH2+,
Fe(OH)2+ , Fe(OH)4-, Fe2(OH)24+,
Fe3(OH)45+, Fe(OH)3(s).
-
Reactions:
H2O <=> H+ + OH-
Log K= -14.0
FeOH2+<==> Fe+3 + OH-
Log K= -11.8
Fe(OH)2+ <==> Fe+3 + 2 OH-
Log K= -22.3
Fe(OH)4- <==> Fe+3 + 4 OH-
Log K= -34.4
Fe2(OH)24+<==> 2 Fe+3
+
2 OH-
Log K= -25.1
Fe3(OH)45+<==>3 Fe+3
+ 4
OH-
Log K= -49.7
Fe(OH)3 (s) <==> Fe+3 + 3 OH-
Log K= -38.8
-
Tableau
and logC-pH diagram (see your book)
-
For the
logC-pH diagram of this system click here
-
For the
MATLAB code used to create this graph, click here.
Example Problem
Add
10-4 M FeCl3 to water at pH 7. Will a precipitate
form? What will be the dominant species in the aqueous phase? What is
the
pH of the solution?
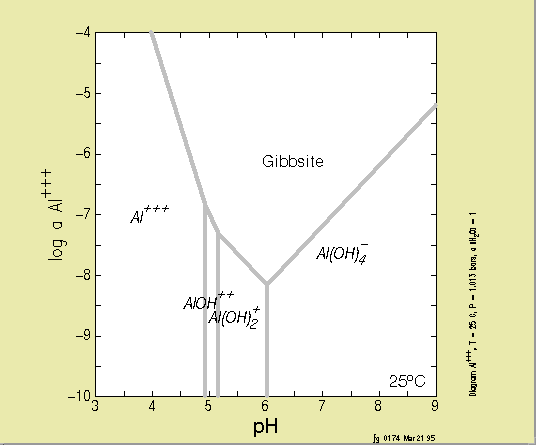
SOLUBILITY DIAGRAMS
Below, you will find a few solubility diagrams. Test yourself to find
out
if you can interpret them!
The first two are solubility diagrams for iron-oxides. What has
changed?
Why?
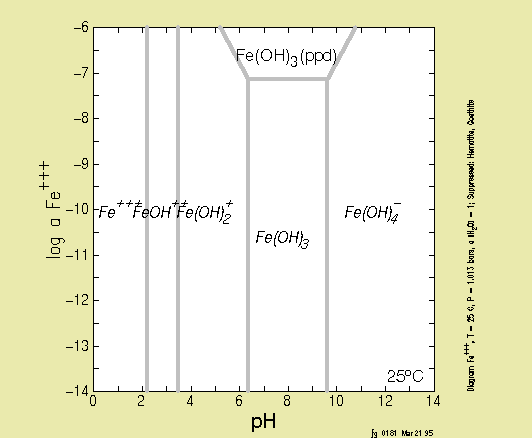
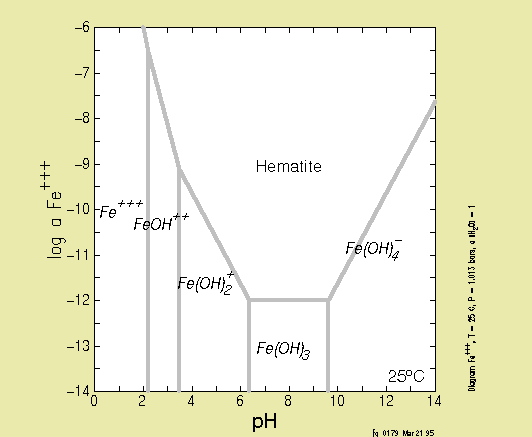
This
is
a solubility diagram for Aluminium